- Introduction
Polyacrylonitrile and its copolymers are among the most widely used polymer materials for preparation of ultrafiltration and microfiltration (MF) membranes. The classic technique used for their synthesis is phase inversion. The development of this method was preceded bysystematic research work major part of which were related to the influences of various parameters on the mechanism and the establishment of the regularities determining membrane morphology[1,p.512, 2,p.231]. It is hard to pre-set and theoretically characterize the resulting membrane structure by taking into account the set of different parameters of the process.In such cases, the relationship is studied individually and it is specific for the particular system.To prepare PAN membranes by the method of phase inversion, solutions of PAN in various solvents and at various ratios are used. Thus, membranes of different technological characteristics and different applications are obtained. The most widely used solvents are dimethylacetamide, N,N – dimethylformamide(DMF), etc. Dimethylsulfoxide is highly polar aprotic solvent having excellent solvent ability for PAN and it is used alone or with additives to prepare mainly hollow fiber membranes [3,p.6]. There are well-known publications reporting for studies on the interactions and peculiarities in DMSO behavior towards organic and inorganic compounds [4,p.7, 5,p.6].
Based on the available literature, it can be stated that the reason is in the type of the interaction between DMSO and the different compounds and their functional groups which affects the membrane formation by phase inversion.
The aim of the present work is to obtain flat asymmetric ultrafiltration membranes by semi-dry phase inversion method from solutions of PAN and DMSO.
- Experimental
Solutions of PAN and DMSO were used for the experiments. They were prepared from polyacrylonitrile fibers obtained from ternary copolymer with composition: acrylonitrile, methacrylate and 2-acrylamide-2-methylpropane sulfonic acid at ratio 90.6 : 8.1 : 1, product of “Lukoil NeftochimBurgas” Co and solvent — DMSO,puriss. p.a., ACSreagent, ≥99.9% (GC), product of „Fluka”, Switzerland.
The membranes were formed by laying a film of the polymer solution onto a substrate of two-side calendered polyester matt of grade FO-2403, product of ,,VelidonFilter”, Germany.
The studies on the transport and selective characteristics of the membranes were carried out with a ultrafiltration laboratory cell SM-165-26 of “Sartorius”, England. The calibrant used for their characterization was AlbuminfractionV (frombovineserum), Мм=76 000 –product of „Merck”, Germany. Selectivity of the different membranes was determined by studying of the resulting filtrate on a UV-VIS spectrophotometer “UNICAM 8625” at the wavelength (λ)=280 nm(foralbumin).The membrane structure formed was visualized by scanning electron microscopy by cathodesputteringwithmicroscopeJSM-5510, product of “JEOL”,Japan.
To prove the physicochemical interactions between the polymer and the solvent, samples of the working solutions were studied by FT-IR spectrophotometer ,,TENSOR 27”, ‘’Bruker”,Germany. The samples were brought as a capillary layer onto KBr tablets. The spectra were registered under the following conditions:interval studied — 3996÷399 cm-1and resolution – 2.
The rheological properties of the polymer solutions were studied by measuring the dynamic viscosity on a rotationalviscosimetertype — REOTESTII -2.1, Germany.
The membranes were prepared by the semi-dry phase inversion method under normal conditions – Т=25 оCand atmospheric pressure in precipitation medium – deionized water with the same temperature. Deionized water was used also for their subsequent washing, thermal treatmentand characterization.
The thermal treatment was carried out at 80 оCfor 10 min with the membranes fixed on a metal frame.
- Results and discussion
The solvent used DMSO, due to its good solvation properties and specific properties, in this case also determined the structural arrangement in the PAN/DMSO solution. This was expected to affect the conditions of the phase inversion process and to be of certain importance for the formation of the membrane structure. For all phase inversions carried out in present work, the conditions were the same except for the polymer solutions concentrations.
The existence of interaction between the solvent and the polymer was shown by the analyses of the solutions with IR spectroscopy.The spectral data for a solution of PAN/DMSOwere compared to these of the pure solvents(Fig.1). Substantial decrease of the intensity of the band at 1026 cm-1,characterizing the O=S group was observed in the PAN/DMSO compared with the spectrum of DMSO. These changes were probably due to coordination interactions with PAN functional groups or other intermolecular interactions [6, p.10].
The degree of these interactions practically affects the viscosity (η)of the system. The structural organization in the solutions is directly connected with their viscosity determined as a result from the influence of the tangential shear stress. The behavior and the rheology of the solutions under mechanical impact (τ) were different.It can be seen from the logarithmic dependence shown in Fig.2, that under the same conditions, the flow curve of the 18 mass% PAN/DMSO solution was the highest standing while that of the 15 mass% PAN/DMSO solution – the lowest.
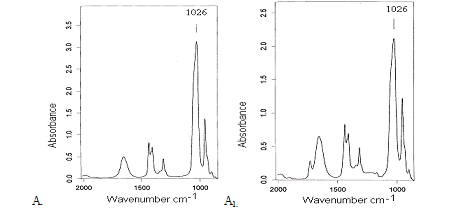
Fig. 1.IRspectra of samples: DMSO (A), PAN/DMSO (A1).
The shapes of the three curves indicate for the stability of the polymer solutions where there is no additional supramolecular structuring, i.e. they are homogeneous under normal temperatures.Accordingly, the structural organization of 18 mass% PAN/DMSOhad the highest viscosity, higher internal friction and smaller rheology. This is due to the differences in the physicochemical behavior of the solvent because of the different amounts of polymer in the solution.The dynamic viscosity measurements and IR spectra showed that the solutions used were stable and homogeneous which are conditions necessary for the phase inversion to take place and the formation of the membrane structure. The studies showed also that solutions with concentrations in the range from 15mass% to 18mass% PANwere the most suitable for formation of UF membranes. Outside this range, the membrane structures did not possess characteristics corresponding to the ultrafiltration scope.
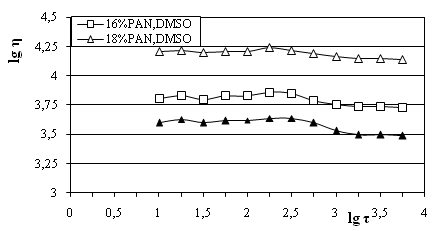
Fig. 2.Flow curves of polymer solutions with different concentration of PAN and DMSO.
The membranes obtained were estimated by studying their performance characteristics – permeability (G, l/m2h)and selectivity (φ, %). The results obtained for membranes formed from solutions with PAN concentrations 15, 16and18mass% with solvent DMSO. The dependencies of the water permeability (GH2O, l/m2h)on pressure (P, MPa) measured in both directions are presented in Fig.3.
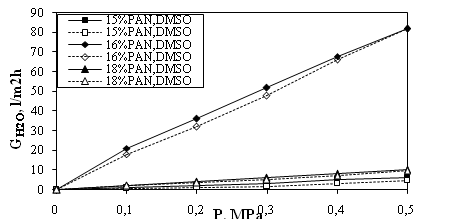
Fig. 3.Hysteresis curves of membranes prepared from solutions of different concentrations of PAN and DMSO.
The effects of pressure and concentration can be read from the hysteresis in Fig.3. The highest water permeability was observed for 16mass% PAN and reached 80 l/m2hat 0.5MPa. The hysteresis area was very small for all the three membranes and it indicated for their mechanical strength in operation but also for greater densification of the polymer structure.
Considering the results shown in Table 1 on the selectivity (φalb) and permeability (Galb, l/m2h) of the membranes under pressure of 0.3 MPa vs the calibrant albumin, it can be seen that again the membrane prepared from 16 mass% PAN solution showed three times higher selectivity for albumin compared to the other two membranes which showed also very low permeability.
Table 1.
Permeability and selectivity for water and albumin of membranes prepared from the corresponding solutions at pressure of 0.3 MPa
| Polymer solution | GH2O,
l/m2h |
Galb,
l/m2h |
φalb,
% |
| 15 mass % PAN/DMSO | 5 | 5 | 21 |
| 16 mass % PAN/DMSO | 24 | 7 | 76 |
| 18 mass % PAN/DMSO | 6 | 4 | 23 |
SEM indeed showed that the selective layer surface was substantially densified and took up comparatively deeper into the transport sublayer and the asymmetry of the structure could be clearly distinguished.

Fig. 4.SEM micrographsof a cross sectionof a membrane formed from solution of 16 mass % PAN/DMSO.
CONCLUSIONS
- The optimal concentration range for preparation of asymmetric membranes was found to be from 15 mass% to 18mass% PAN/DMSO.
- It was established that, as a result from the intermolecular interactions between PAN and DMSO, the best ultrafiltration properties had the membrane prepared from 16 mass%PAN/DMSO.
- The membrane formed from the solution of 16 mass% PAN/DMSO had low permeability for albumin as a result from the densification of the surface layer.
REFERENCES
- Мулдер М., Введение в мембранную технологию, Москва, Изд. Мир, (1999), с. 512
- Дубяга В.П., Перепечкин Л.П., Каталевский Е.Е., Полимерные мембраны, Москва,Изд.”Химия”(1981), с.231.
- TanL., LiuS., Pan D., Water effect on the gelation behavior of polyacrylonitrile/dimethyl sulfoxide solution, Colloids and Surfaces A: Physicochem. Eng. Aspects, (2009), Vol.340, р. 168–173.
- Tan L., Pan D., Pan N., Gelation behavior of polyacrylonitrile solution in relation to aging process and gel concentration, Polymer, (2008), Vol.49, p.5676-5682.
- Du W., ChenH., XuH., PanD., Pan N.,Viscoelastic behavior of polyacrylonitrile/dimethyl sulfoxide concentrated solution with water, Journal of Polymer Science Part B: Polymer Physics, (2009),Vol.47, p.1437–1442.
- Wu Q.Y., Chen X.N., Wan L.S., Xu Z.K., Polyacrylonitrile and Solvents: Density Functional Theory Study and Two-Dimensional Infrared Correlation Analysis, Journal ofPhysical Chemistry B, (2012), Vol.116, p.8321-8330.[schema type=»book» name=»PREPARATION OF FLAT ASYMMETRIC MEMBRANES FROM SOLUTIONS OF PAN AND DMSO » description=»The possibility to obtain flat asymmetric ultrafiltration (UF) membranes by phase inversion from solutions of polyacrylonitrile (PAN) and dimethylsulfoxide (DMSO) was studied. The physicochemical interactions between the polymer and the solvent were studied by infrared spectroscopy and dynamic viscosity measurements. According to the transport, selective and microscopic characteristics determined, the best properties showed the membrane prepared from 16 mass % PAN/DMSO.» author=»Miteva Milena Pencheva, PetrovStoikoPetrov, TodorovaDonkaDimitrova» publisher=»БАСАРАНВОИЧ ЕКАТЕРИНА» pubdate=»2017-03-29″ edition=»ЕВРАЗИЙСКИЙ СОЮЗ УЧЕНЫХ_30.04.2015_04(13)» ebook=»yes» ]

