The diagnosis of pleural tumors, despite the progress in clinical and morphological methods, is still difficult. In this anatomic region develop a few primary tumors, and the most common of them is the malignant pleural mesothelioma (MPM). This is also the location of numerous metastases from different malignant tumors of various histogenesis. This diversity of malignant tumors requires first of all to differentiate them from the pleural mesothelioma – a tumor of high malignancy and unfavourable prognosis. Its difficult histological diagnosis is confirmed either rarely in life with reliable electronic microscopic study or after death with an autopsy. The uncertain imaging diagnostics and the difficult interpretation of cytological material from the pleura require a necessary immunehistochemical study of tissue biopsy material. [1, p. 24-27]
Malignant mesothelioma is a rare, asbestos-associated tumor, which originates from the pleural mesothelium, peritoneum, pericardium and tunica vaginalis of testis. Its frequency is less than 1% of all malignancies. It most commonly affects the pleura (80-90%), the peritoneal cavity (10-20%) and there are only isolated cases of other locations. [8. pp. 1-11], [7 p. 395]
The aim of this study was to adopt into practice an optimized immunohistochemical panel for correct diagnosis of MPM.
Material and Methods
Tumor Samples
We conducted a study on 50 mesotheliomas diagnosed over a 6-year period in the Department of Clinical Pathology at St George University Hospital.
The distribution of malignant mesotheliomas by histological type is shown in Figure 1. According to the WHO histological classification for 2004, it includes the following types of malignant mesothelioma:
- epithelioid – 34 (64.0%)
- biphasic – 13 (26.0%)
- sarcomatoid – 3 (8.0%)
- desmoplastic – 1 (2.0%)
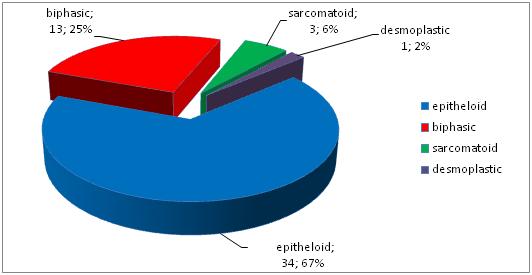
Fig. 1 Histological types of malignant pleural mesothelioma
In the immunohistochemical study of biopsy pleural material was used a panel of 10 antibodies from different companies. (see Table 1)
Table 1
Antibodies used in immunohistochemical study
|
Antibody |
Source |
Clone |
Dilution |
Pretreatment |
Expression |
|
Mesothelin |
Leica Biosystems Novocastra Ltd. Newcastle UK |
5B2 ММА* |
RTU** |
High temperature with 0.01М СВ*** (рН 6.0). |
membranous
|
|
WT1 |
Leica Biosystems |
WT49 ММА |
1:100 |
High temperature рН 9.0 for 15 min. | nuclear |
|
Calretinin
|
Leica Biosystems |
5A5 ММА |
RTU |
High temperature with 0.01М СВ (рН 6.0) for 20 min. |
nuclear and cytoplasmic |
|
Anti-D240 antibody |
Abcam Cambridge. UK
|
D240 ММА |
1:40 |
High temperature 95-100оС СВ (рН 6.0), for 20 min. |
membranous |
| ТТF-1
thyroid transcription factor |
Leica Biosystems |
SPT24 ММА |
1:200 |
Water bath 980С with pH 6.0 for 20 min. | nuclear
|
|
Cytokeratin 7 |
Leica Biosystems |
OV-TL 12/30 |
RTU
|
High temperature with 0.01 М citrate buffer рН 6.0. |
cytoplasmic
|
|
Cytokeratin 20 |
Leica Biosystems |
KS20.8 ММА |
RTU
|
High temperature with 0.01 М рН 6.0. |
cytoplasmic |
| ЕМА
Epithelial Membrane Antigen |
Leica Biosystems |
GP1,4 ММА |
RTU |
Protease |
cytoplasmic and membranous |
| CEA carcinoembyonic antigen |
Leica Biosystems |
12-140-10 ММА |
1:200 |
Protease |
cytoplasmic |
|
Cytokeratin 5/6
|
DАКО. Agilent Technologies соmр. Denmark |
D5/16 B4 ММА |
1:100 |
100° C for 20 min in 10 mmol/L Tris buffer pH 9.0. |
cytoplasmic |
*ММА – mouse monoclonal antibody
**RTU – ready to use
***СВ – citrate buffer
Immunohistochemical Analysis This panel was applied to all 50 mesotheliomas, which included three main histological types – epithelioid, biphasic and sacromatoid.
The aim of this study was to determine the level of expression for the different histological mesotheliomas and then compare them to those of metastatic lung adenocarcinomas. For that reason it was necessary to appraise the specificity and sensitivity of each of them and in accordance with the obtained results to select the most suitable antibodies to use in the diagnosis of malignant mesotheliomas and metastatic pleural carcinomas. Therefore was developed a rating scale for the intensity of reaction colour and percentage of cells with positive expression. The intensity of reaction colour adopted the following values: 0 – missing; 1 – weak; 2 – moderate; 3 – strong. Strong intensity is considered the reaction colour of the positive control. The percentage of positively stained tumor cells was determined as follows: 0 – 10%=0; 10 – 39%=1; 40 – 69%=2; 70 – 100%=3. The sum of the numerical values of the two indexes ranged from 0 to a maximum value of 6. This allowed us to appraise in more detail the weak positive and focal staining reactions, which expressed the presence of the respective tissue antigen. Reading the respective expression in the nucleus, cytoplasm or cell membrane took into consideration the expected results, specified in the protocols for the respective antibodies of the company producer. All the cases with expression of more than 10%, were accepted as positive. For the statistical analysis and in accordance with the above scales, the tumors can be divided into three groups:
- Negative (–) ● all tumors with no colour reaction or which is up to 10% of the tumor cells.
- Weak positive (+) ● with sum of the two indexes from 2 to 4
- Strong positive (++) ● from 5 to 6. See Table 2
Interpretation of Results
Table 2
Immunohistochemical study of different histologic variants of МPM
|
Antibody |
Epithelioid mesothelioma n=32 |
Biphasic mesothelioma n=13 |
Sarcomatoid mesothelioma n=5 |
Mesotheliomas total n=50 |
Mesotheliomas total n=50 |
|||||||||
| group | group | Group | group | group | group | |||||||||
| 1* | 2* | 3* | 1 | 2 | 3 | 1 | 2 | 3 | 1 | 2 | 3 | (+/-) | %р | |
| (–) | (+) | (++) | (–) | (+) | (++) | (–) | (+) | (++) | (–) | (+) | (++) | |||
| CK 5/6 | 2 | 4 | 26 | 0 | 4 | 9 | 3 | 1 | 1 | 5 | 9 | 36 | 45/5 | 90% |
| D2-40 | 0 | 5 | 27 | 1 | 5 | 7 | 1 | 3 | 1 | 2 | 13 | 35 | 48/2 | 96% |
| Calretinin | 2 | 4 | 26 | 1 | 3 | 9 | 3 | 1 | 1 | 6 | 8 | 36 | 44/6 | 88% |
| Mesothelin | 6 | 5 | 21 | 3 | 4 | 6 | 5 | 0 | 0 | 14 | 9 | 27 | 36/14 | 72% |
| WT1 | 4 | 8 | 20 | 3 | 3 | 7 | 3 | 1 | 1 | 10 | 12 | 28 | 40/10 | 80% |
| ЕМА | 11 | 8 | 13 | 4 | 5 | 4 | 3 | 1 | 1 | 18 | 14 | 18 | 32/18 | 64% |
| CK 7 | 14 | 4 | 14 | 2 | 4 | 7 | 3 | 2 | 0 | 19 | 10 | 21 | 31/19 | 62% |
| CK 20 | 28 | 3 | 1 | 13 | 0 | 0 | 5 | 0 | 0 | 46 | 3 | 1 | 4/46 | 8% |
| CEA | 32 | 0 | 0 | 11 | 2 | 0 | 5 | 0 | 0 | 48 | 2 | 0 | 2/48 | 4% |
| TTF1 | 28 | 1 | 0 | 15 | 0 | 0 | 5 | 0 | 0 | 49 | 4 | 0 | 1/49 | 2% |
1*– negative; 2*– weak positive; 3* – strong positive
With the different histological types of МPM, the percentage distribution of the positive results for the used antibodies was highest for D2-40 (96%), CK 5/6 (90%) and Calretinin (88%). The next in order were WT1 (80%), Mesothelin (72%), reaching the lowest level of expression with TTF1 – 2%.
Table 3 shows the summarized results, obtained from the expression of the same antibodies, which only accounted for the presence or absence of expression, and as positive also accepted all the cases with intensity of colour higher than 10%.
Table 3
Percentage of positive expression (%p), sensitivity and specificity for mesotheliomas and metastatic lung adenocarcinomas
|
Antibody |
Epithelioid and biphasic mesothelioma n=45 |
Metastatic tumors |
Sensitivity and specificity
|
|||
| Lung adenocarcinoma
n=41
|
Sensitivity
|
Specificity |
||||
| +/– | %p | +/– | %p | % | % | |
|
Antibodies positive for mesothelioma |
||||||
| CK 5/6 | 43/2 | 95.55 | 9/32 | 21.95 | 95.55 | 78.04 |
| D2-40 | 44/1 | 97.77 | 2/39 | 4.87 | 97.77 | 95.12 |
| Calretinin | 42/3 | 93.33 | 4/37 | 9.75 | 93.33 | 90.24 |
| Mesothelin | 36/9 | 80 | 4/37 | 9.75 | 80 | 90.24 |
| WT1 | 38/7 | 84.44 | 8/33 | 19.51 | 84.44 | 80.48 |
| ЕМА | 30/15 | 66.66 | 32/9 | 78.04 | 66.66 | 21.95 |
|
Antibodies positive for adenocarcinomas |
||||||
| CK 7 | 25/20 | 55.55 | 35/6 | 85.36 | 85.36 | 44.44 |
| СК 20 | 4/41 | 8.88 | 6/35 | 14.63 | 14.63 | 91.11 |
| CEA | 2/43 | 4.44 | 33/7 | 24.39 | 80.48 | 95.55 |
| TTF1 | 1/44 | 6.66 | 40/1 | 97.56 | 97.56 | 97.77 |
Note. Due to their specific histological picture, the sacromatoid mesotheliomas were taken out of the study in order to distinguish MPM from the metastatic lung adenocarcinomas.
To appraise the sensitivity and specificity of the markers used in the differential diagnosis of MPM and pleural metastases from lung adenocarcinomas, the standard 2х2 table was used. The markers used were divided into two groups – positive for mesothelioma and positive for metastatic adenocarcinomas.
With mesotheliomas, from the positive markers the highest sensitivity and specificity were displayed by D2-40 and Calretinin, followed by Mesothelin and WT1. СК 5/6 had a very high sensitivity, but lower specificity, whereas Mesothelin had low sensitivity and high specificity, which makes it a suitable ancillary marker in confirming the mesothelial origin of the tumor.
With the metastatic lung adenocarcinomas TTF1 had very high sensitivity and specificity, followed CEA. СК7 had good sensitivity but low specificity. Figure 2.

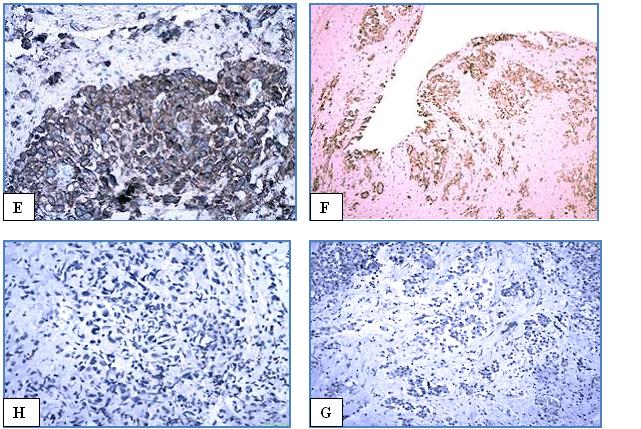
Epithelioid and biphasic mesothelioma and lung adenocarcinoma
Fig. 2 IHC study of epithelioid mesothelioma
A – НЕ x100.
Positive markers; B – D2-40. x100; C – CK 5/6 x100; D –mesothelin x100; E – calretinin x200 ; F – WT1 x100;
Negative markers: H – TTF1. x100; G – CEA x100
Discussion:
With its wide differential diagnosis, difficult therapy and unfavourable prognosis, the accurate diagnosis of the malignant mesothelioma nowadays is crucial for the treatment of the patient. [3, p. 1-11]
The histological picture of the malignant mesothelioma and the metastatic tumors in most part are very similar and are often indistinguishable in routine colouring with hematoxylin and eosin. Is particularly refers to metastases of lung adenocarcinomas. The only way to a correct diagnosis of pleural tumor remains the immunohistochemical study of biopsy material. There are great difficulties in defining a unique reproductive immunophonotype of MPM, which we can use in differential diagnosis with metastatic pleural tumors. [9, p. 1316]
The immunohistochemical study (ICH) has existed for more than 25 years. In the diagnostic practice for that time were offered various antibodies and combinations of them, but still the “gold standard“ for this study has not been found yet. Today it plays an important part in diagnosing MPM. Without it the differential diagnosis of this kind of tumor is most of the times impossible. The literature has described dozens of bigger and smaller antibody panels, but none of them has proven entirely accurate and efficient for a correct morphological diagnosis. That’s why most of the authors today suggest big panels of 10-12 antibodies, which makes the immunohistochemical study significantly more expensive. [11 pp. 1-19]
There are many reasons for these, sometimes significant differences in the obtained results from the immunohistochemical study, but the main one is the phenotypical flexibility of the mesothelium, typical for the embryonic differentiation of the mesoderm. Second are the objectively limited possibilities of the immunohistochemical method itself. Today there are dozens of companies offering different antibodies with different sensitivity and specificity. Despite the existing automated systems for IHC study, the records for processing the material are still too cumbersome and bulky. We shouldn’t also forget the very important subjective factor in assessing the results from this study. [12, p. 253]
The lack of absolutely specific and sensitive immunohistochemical markers for mesothelioma makes the differential diagnosis of this tumor very difficult. It depends entirely on using the immunohistochemical panel comprised of markers positive for mesothelioma (i.e. markers which are most often expressed in mesothelioma, but not in carcinomas) and epithelial markers negative for mesothelioma (i.e. markers which are most often expressed in carcinomas, but not in mesotheliomas). The recommended panels constantly change as a result of identifying new antibodies, which can be used in the differential diagnosis of these tumors and of the new information about the diagnostic value of the individual markers [2, p. 397], [12, p. 253]
The obtained results showed that highest sensitivity and specificity as positive markers for MPM were manifested by D2-40 and Calretinin, followed by Mesothelin and WT1. СК 5/6 had a very high sensitivity, but lower specificity, whereas Mesothelin had low sensitivity and high specificity, which makes it a suitable ancillary marker in confirming the mesothelial origin of the tumor.
ЕМА and СК7 had low specificity, which showed that they are expressed with very close results, in both МРМ and the metastatic lung adenocarcinomas. That’s why they are not suitable for differential-diagnostic search in primary and metastatic pleural tumors. These results are close to those reported by Husain AN et al. 2013 and Ordóñez NG 2013, but different from the data of King JE et al. in 2006, which tells of an evolution in specifying the antibody panel to determine the immunophenotype of MPM. [4, p. 647], [11, pp. 1-19], [5, p. 223]
In the differential diagnosis we considered:
The number of antibodies used for this purpose today is too large. [6, p. 190] Considering the sensitivity and specificity of the antibodies that we investigated in our study, the recommended positive markers for epithelioid mesothelioma with the highest sensitivity and specificity were D2-40 and Calretinin. For the metastases of lung adenocarcinomas the best sensitivity / specificity ratio was for TTF-1 and СЕА.
Conclusion
IHC testing is essential for the diagnosis and differential diagnosis of MPM with metastatic pleural adenocarcinomas. It must necessarily include a panel of positive and negative (carcinoma-related) markers.
The suitable optimal combination to distinguish the malignant mesothelioma from metastatic lung adenocarcinomas should be a panel of antibodies with the highest possible sensitivity and specificity. This gives us reason to recommend an economically optimized immunohistochemical panel of two antibodies positive for mesothelioma: D2-40 and Calretininи, and two negative, but positive for lung adenocarcinomas: TTF1 and СЕА.
References
- Костов К, Димитър К. Плеврата. изд. Сиела. София 2006:24-27
- Alberto MM. Application of Immunohistochemistry to the Diagnosis of Malignant Mesothelioma. Arch Pathol Lab Med 2008;132:397-401
- Galateau-Sallé F. Pathology of Malignant Mesothelioma Springer-Verlag London Limited 2006:1-11
- Husain AN., ThomasVC., Nelson GO., et al. Guidelines for Pathologic Diagnosis of Malignant Mesothelioma 2012 Update of the Consensus Statement from the International Mesothelioma Interest Group. Arch Pathol Lab Med 2013;137:647-667
- King JE, Thatcher N, Pickering CA at al. Sensitivity and specificity of immunohistochemical markers used in the diagnosis of epithelioid mesothelioma: a detailed systematic analysis using published data. Histopathology 2006;48: 223–232
- Kushitani K, Takeshima Y, Amatya VJ, Furonaka O, Sakatani A, Inai K. Immunohistochemical marker panels for distinguishing between epithelioid mesothelioma and lung adenocarcinoma. Pathol Int. 2007;57:190–199
- Loggie BW. Malignant peritoneal mesothelioma. CurrTreat Options Oncol. 2001;2:395–399.
- Moore AJ., Robert JP., John W. Malignant mesothelioma. Orphanet Journal of Rare Diseases 2008;(3)34:1-11
- Mani H. Dani SZ. Immunohistochemistry. Applications to the Evaluation of Lung and Pleural Neoplasms: Part 1. Chest 2012; 142:(5):1316-1323
- Ordonez NG., Immunohistochemical Diagnosis of Epithelioid Mesothelioma An Update. Arch Pathol Lab Med. 2013;129:1407-1414
- Ordóñez NG. Application of immunohistochemistry in the diagnosis of epithelioid mesothelioma: a review and update. Human Pdthology 2013;44(1):1-19
- Roberts F, Harper CM, Downie I, Burnett RA. Immunohistochemical analysis still has a limited role in the diagnosis of malignant mesothelioma: a study of thirteen antibodies. Am J Clin Pathol. 2009;116:253–262[schema type=»book» name=»DIAGNOSTIC VALUE OF THE IMMUNOHISTOCHEMICAL STUDY FOR MALIGNANT PLEURAL MESOTHELIOMA AND LUNG ADENOCARCINOMAS» author=»Peshev Zhivko Vladimirov, Belovezhdov Veselin Todorov, Novakov Ivan Petkov» publisher=»БАСАРАНОВИЧ ЕКАТЕРИНА» pubdate=»2017-04-04″ edition=»ЕВРАЗИЙСКИЙ СОЮЗ УЧЕНЫХ_30.04.2015_4(13)» ebook=»yes» ]

