Introduction
Endometriosis is a common benign gynecologic pathology defined as an ectopic localization of endometrial tissue. It can affect any woman from promenarche to postmenopausal period.
Endometriosis is divided in two types according to the localization: endopelvic and extrapelvic [16, p. 53]. Within the abdomen, endometriosis can be classified as intraperitoneal or extraperitoneal. When located 5 mm under the peritoneal surface, it is defined as deep infiltrating endometriosis. Endometriosis affects most commonly the ovaries (30%), the utero-sacral and large ligaments (18%-24%), the fallopian tubes (20%), the pelvic peritoneum, Douglas’ pouch, the appendix and the small and large intestines [7, p. 6677]. According to the pathological reports, the incidence of soft tissue endometriosis is 2.8%, of gastrointestinal endometriosis is 0.3%, and of the urinary tract- 0.2% [8, p. 53]. Endometriosis rarely affects extraabdominal organs, such as the lungs, urinary system, skin and central nervous system [6, p. 3431].
Endometriosis is the most common cause of chronic pelvic pain in females. 60% to 70% of the cases with chronic pelvic pain are due to endometriosis [7, p. 6676]. It is more common (15-25%) among women with infertility problems and is extremely rare after menopause, because of the estrogen dependence of the ectopic tissue [4, p. 442].
Endometriosis is not related with endometrial cancer. Some researches have demonstrated a relationship between endometriosis and ovarian cancer, non-Hodgkin lymphoma and brain cancer [14, p. 332]. Concerning ovarian cancer, endometriosis is reported to be significantly more frequent in women with endometrioid and clear cell carcinomas, than in those with other subtypes of ovarian carcinoma [15, p. 1156]. Atypical endometriosis is a precursor lesion for endometrioid and clear cell ovarian carcinomas.
There are three theories for the pathogenesis of endometriosis: theory of metaplasia, theory of dispersion of the endometrium and theory of induction. The first theory was proposed by Meyer in 1903 and refers to metaplasia of the peritoneal serosa [3, p. 37]. According to this theory, the development of endometriosis is a result of inflammatory or hormonal factors, which cause tissue differentiation of mesothelial cells of the peritoneum in endometrial glands and stroma. This theory can explain the development of endometriosis in distant organs. The second theory is that of endometrium dispersion- during the menstruation, retrograde dispersion of living endometrial cells can occur in the peritoneal cavity and ovaries. The third theory is that of induction. According to this theory, unknown substances are released from the endometrium and cause transformation of undifferentiated mesenchymal cells to endometrial tissue [9, p. 194].
Case report
44 years old woman presented with chronic pelvic pain and chronic obstipation was firstly admitted to the Department of Obstetrics and Gynecology and later- to the Surgical department of Pulmed Hospital- Plovdiv. During the performed surgical intervention a consultation with a surgeon was needed prior to tumor formation of the rectum. The intervention included rectal resection and termino-terminal anastomosis. The patient had a history of supravaginal hysterectomy 4 years ago.
Materials and methods
The specimens were fixed in 10% formalin and embedded in paraffin. Serial sections cut from the paraffin-embedded blocks were used for routine histopathology with Hematoxylin-Eosin staining and immunohistochemically analyzed for the expression of E-cadherin.
Results
The macroscopic examination revealed 4cm in diameter cystic tumor formation in the rectal wall with hemorrhagic content (Fig.1).
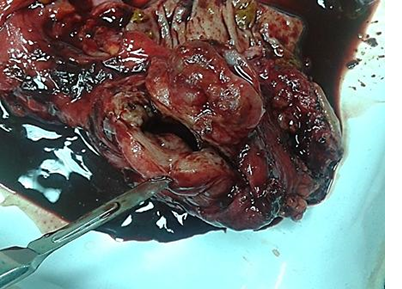
Fig. 1. Endometrial cyst with hemorrhagic content inside the rectal wall.
The histologic examination showed rectal endometriosis- cystic degeneration with presence of endometrial glands and stroma inside the rectal wall (Fig.2).
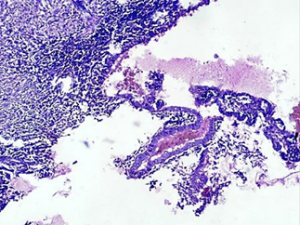
Fig. 2. Endometriosis with cystic degeneration inside the rectal wall, H-E, magn. х40.
The immuohistochemical E- cadherin expression in the ectopic endometrial glands showed 1+ (low-intensity immunoreactivity of more than 10% of the epithelial cells)(Fig. 3).
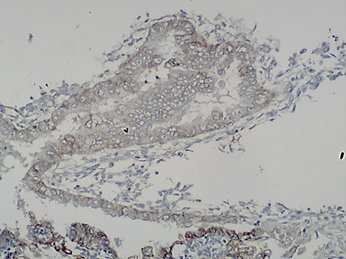
Fig. 3. Immunohistochemical E-cadherin expression in the ectopic endometrial glands, magn. х100: 1+ (low-intensity immunoreactivity of more than 10% of the epithelial cells).
Discussion
Endometriosis is an estrogen-dependent inflammatory dysfunction first described by Rokitansky in 1860 and characterized by the presence of glands and stroma that histologically resemble functional endometrial tissue but are located outside of the uterus [11, p. 431].
This condition shares a number of features with cancer such as its stem cell-like activity, the ability to evade apoptosis and to induce angiogenesis, as well as the intrinsic self-regulation of its rate of proliferation. Such properties can be explained through the molecular signals between the cells which regulate the intracellular signaling cascades. This allows the population of ectopic cells to survive within its new tissue environment [12, p. 579].
Endometrium is derived from intermediate mesoderm via mesenchymal-epithelial transition during the development of the urogenital system. By retaining some imprint of their mesenchymal origin, endometrial epithelial cells may be able to return to this state, via epithelial-mesenchymal transition [10, p. 718]. To facilitate its attachment, endometrial tissue can induce epithelial-mesenchymal transitions in mesothelial cells. The capacity of the endometrial tissue to invade and acquire a blood supply contributes to the implantation process. After implantation, local production of estrogens provides continuous, ovary-independent growth stimulus [1, p. 238].
For endometriosis, studies on E-cadherin expression have led to contradictory results. While some studies reported a reduction of E-cadherin expression in endometriosis compared with the endometrium [5, p. 265], others found no difference in E-cadherin expression in endometriosis compared with the endometrium [2, p. 878]. In vitro, E-cadherin-negative, N-cadherin-positive endometriotic epithelial cells showed invasive growth [13, p. 75].
Matsuzaki S. et al. hypothesized that epithelial cells of pelvic endometriosis originate from retrograde menstruation of endometrial epithelial cells and the epithelial-mesenchymal and mesenchymal-epithelial transiyions might be involved in the pathogenesis of pelvic endometriosis. They investigated endometriosis with different localizations, including deep infiltrating endometriosis (localized in the rectovaginal septum, uterosacral ligament, bladder wall or bowel wall). Their study demonstrated that E-cadherin expression was significantly higher in epithelial cells of deep infiltrating endometriosis compared with those of menstrual endometrium. These findings suggest that mesenchymal-epithelial transition might occur in deep infiltrating endometriosis [10, p. 718].
The presented case is particularly interesting because of the unusual location of the endometrial implant, mimicking tumor of the rectum. The immunohistochemical E-cadherin in the presented case is 1+ (low-intensity immunoreactivity of more than 10% of the epithelial cells). This result may be explaned with the process of epithelial-mesenchymal transition and may suggest invasive growth of the endometriosis.
Conclusion
The diagnosis of gastrointestinal endometriosis can be challenging. The clinical presentations can vary widely, depending on the localization and mimicking tumor processes. Because endometriosis is a benign process with the ability to metastasize, the epithelial- mesenchymal transition can be involved in its pathogenesis.
References
- Annemiek W., Nap M.D., Patrick G. et al. Pathogenesis of endometriosis. Best Practice & Research Clinical Obstetrics and Gynaecology Vol. 18, No. 2, pp. 233–244, 2004.
- Bartley J., Julicher A., Hotz B. et al. Epithelial -mesenchymal transition (EMT) seems to be regulated differently in endometriosis and the endometrium. Arch Gynecol Obstet. 2014;289(4):871–81.
- Bischoff F.Z., Simpson J.L. Heritability and molecular genetic studies of endometriosis. Hum Reprod Update 2000, 6:37–44.
- Bulletti C, Coccia ME, Battistoni S, Borini A: Endometriosis and infertility. J Assist Reprod Genet 2010, 27:441–447).
- Chen Y.J., Li H.Y., Huang C.H. et al. Oestrogen-induced epithelial-mesenchymal transition of endometrial epithelial cells contributes to the development of adenomyosis. J Pathol. 2010;222(3):261–70.
- De Ceglie A., Bilardi C., Blanchi S. et al. Acute small bowel obstruction caused by endometriosis: a case report and review of the literature. World J Gastroenterol 2008; 14: 3430-3434.
- Insilla A. C., Granai M., Gallippi P. et al. Deep endometriosis with pericolic lymph node involvement: A case report and literature review. World J Gastroenterol 2014 June 7; 20(21): 6675-6679.
- Lee H. J., Park Y. M., Jee B. C. et al. Various anatomic locations of surgically proven endometriosis: A single-center experience. Obstet Gynecol Sci 2015;58(1):53-58.
- Machairiotis N., Stylianaki A., Dryllis G. et al. Extrapelvic endometriosis: a rare entity or an under diagnosed condition? Diagnostic Pathology 2013, 8:194.
- Matsuzaki S., Darcha C. Epithelial to mesenchymal transition-like and mesenchymal to epithelial transition-like processes might be involved in the pathogenesis of pelvic endometriosis. Human Reproduction, Vol.27, No.3 pp. 712–721, 2012.
- Nezhat C., Hajhosseini B., King L.P. Laparoscopic management of bowel endometriosis: predictors of severe disease and recurrence. JSLS 2011; 15: 431-438.
- Pollacco J., Sacco K., Portelli M. et al. Molecular links between endometriosis and cancer. Gynecological Endocrinology, 2012; 28(8): 577–581.
- Proestling K., Birner P., Gamper S. et al. Enhanced epithelial to mesenchymal transition (EMT) and upregulated MYC in ectopic lesions contribute independently to endometriosis. Reproductive Biology and Endocrinology (2015) 13:75.
- Somigliana E., Vigano P., Parazzini F. et al. Association between endometriosis and cancer: a comprehensive review and a critical analysis of clinical and epidemiological evidence. Gynecol Oncol 2006, 101:331–341.
- Vercellini P., Scarfone G., Stellato G. et al. Site of origin of epithelial ovarian cancer: the endometriosis connection. British Journal of Obstetrics and Gynaecology. September 2000, Voll 07, pp. 1155-1157.
- Yu J. H. , Lin X. Y., Wang L. et al. Endobronchial endometriosis presenting as central-type lung cancer: a case report. Diagn Pathol 2013, 8:53.[schema type=»book» name=»Deep infiltrating endometriosis mimicking rectal tumor» description=»Endometriosis is a common benign gynecologic pathology defined as an ectopic localization of endometrial tissue. Within the abdomen, endometriosis can be classified as intraperitoneal or extraperitoneal. When located 5 mm under the peritoneal surface, it is defined as deep infiltrating endometriosis. Along with the three theories for the mechanism of development, epithelial-mesenchymal and mesenchymal-epithelial transitions might be involved in the pathogenesis of endometriosis. We present a case of deep infiltrating endometriosis mimicking rectal tumor, with investigation of E-cadherin immunohistochemical expression.» author=»Bozhkova Desislava Milenova, Poriazova Elena Galabova, Karafeziev Velin Rozenov» publisher=»БАСАРАНОВИЧ ЕКАТЕРИНА» pubdate=»2017-01-19″ edition=»ЕВРАЗИЙСКИЙ СОЮЗ УЧЕНЫХ_28.11.15_11(20)» ebook=»yes» ]

